Microbiology automation is an important trend in the industry right now and enrichment and transport media is at the heart of it.
Studies have shown flocked swabs facilitate greater specimen collection and complete elution. The elution of a specimen from such swabs into liquid has demonstrated a significant increase in the release of viable organisms from the swab, which translates into increased sensitivity for detection of microorganisms in the specimen. Consequently, more and more labs are using flocked swabs in combination with a transport media to aid the recovery and detection of pathogens after collection.
Increased specimen sensitivity is not the only benefit. Placing specimens in a liquid based transport allows innoculation and smear preparation to be done with automation platforms. And since automation saves time and money, more and more companies are offering liquid based media to fit the new processors.
But what kind of medium do you need? What are the differences between them? We'll walk you through four of the most common (and important) enrichment and transport media.
Transport Media
A transport medium is a non-nutritive, balanced, buffered medium that provides a controlled environment to maintain the viability of microorganisms without a significant increase in growth from the time of collection to the time it is processed at a lab.
Transport media vary depending on what type of specimen is being collected. They're generally classified on the basis of physical state (semi-solid and liquid) as well as their use in bacterial or viral transport media.
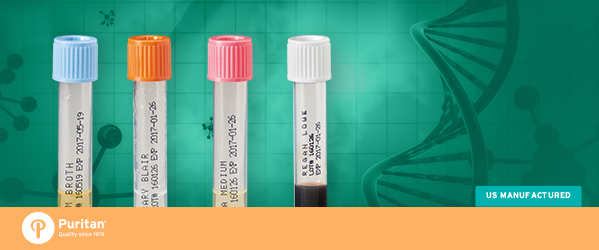
Cary-Blair
In the 1960s Cary and Blair modified Stuart's medium—which consisted of Sodium Thioglycolate, Sodium Glycerophosphate, Calcium Chloride and Methylene Blue as a color indicator—by replacing Sodium Glycerophosphate with inorganic phosphate and raising the pH to 8.4. The resulting medium brilliantly supported the viability of the enteric pathogens (salmonella, listeria, and e. coli) in fecal samples.
The formula has changed little over for the past several decades and Cary-Blair is the preferred transport medium for the preservation of fecal and rectal specimens containing enteric pathogenic bacteria.
Regan-Lowe
Regan and Lowe discovered the medium useful for the selective isolation of B. pertussis and B. parapertussis—the agents that cause whooping cough. Whooping cough is a respiratory infection that can be severe in young infants. Early diagnosis and treatment are essential to limit the progression of the disease and minimize its transmission.
Regan-Lowe transport medium is a selective medium used for transport of nasopharyngeal specimens to the laboratory for qualitative procedures. The base medium contains charcoal to neutralize toxic fatty acids in the specimen. It is supplemented with cephalexin to inhibit bacteria indigenous to the nasopharynx. Horse blood is added to the medium to support the growth of Bordetella species.
Enrichment Media
An enrichment culture is a medium with specific and known qualities that favors the growth of a particular microorganism. The enrichment culture's environment supports the growth of that selected microorganism while inhibiting the growth of others. Growth is promoted by providing an organism with essential nutrients. However rarely, inhibitory substances are added to prevent the growth of normal competitors.
Enrichment media are needed when a low amount of potential pathogens is assumed to be present in a specimen. Those specimens are enriched to multiply the low number.
MRSA
These transport media are used for isolation of Staphylococcus aureus, particularly methicillin-resistant Staphylococcus aureus (MRSA). S. aureus is one of the most common causes of skin and soft tissue infection in both healthcare and community settings.
Enrichment broths are commonly used to increase sensitivity testing for MRSA by increasing isolation rates. Tryptic Soy Broth (TSB) often serves as the base medium to enhance the growth of S. aureus. TSB contains enzymatic digest of casein and enzymatic digest of soybean meal, which provide amino acids and complex nitrogenous compounds that promote microbial growth. Dextrose acts as a carbon energy source that facilitates growth. Dipotassium phosphate acts as a buffering agent. Sodium chloride is added to inhibit or partially inhibit microorganisms other than S. aureus.
LIM Broth
LIM Broth is a selective enrichment broth medium used for the isolation of Group B Streptococcus (GBS). Group B Streptococcus (GBS) is the most common cause of infections such as sepsis, meningitis and pneumonia among newborns. The disease is transmitted to newborns through the mother who carries GBS in her rectum or genital tract during birth.
The CDC suggests using vaginal and rectal swabs with selective enrichment broths to detect GBS colonization from the suspected pregnant women for culture-based screening between 35 and 37 week’s gestation. A LIM Broth's peptones, dextrose, and yeast extract provide nutritional base for growth of GBS. Nalidixic acid and colistin suppress growth of gram-negative bacteria.
This is, of course, only a brief overview of a few important media in clinical microbiology. Custom media systems are also available, which is crucial as automated specimen processors become more popular in labs. Don't hesitate to speak with one of our knowledgeable product specialists today if you're still unsure of what you need. We're here to help!