The quest for fast, accurate wats to detect enteric bacterial DNA for diagnosis and microbiome analysis just took a big step forward.
A recent study confirms that Puritan’s Fecal Opti-Swab® transport system preserves enteric pathogen DNA following prolonged storage without the need for special handling procedures.
What’s the connection?
When an illness is likely caused by an enteric pathogen, confirmation can be quickly made by analysis of a fecal sample for DNA. Swift confirmation helps to ensure that appropriate diagnoses and effective treatments are administered in a timely fashion.
Results Unveiled at ECCMID 2018
Researchers at the University of New England shared the news at the 2018 European Congress of Clinical Microbiology and Infectious Disease (ECCMID) conference in Madrid.
Their study, “Evaluation DNA Recovery of Enteric Pathogens from Puritan® Fecal Opti-Swab Collection and Transport System Following Prolonged Storage,” describes the scientific protocols used to assess Puritan’s Fecal Opti-Swab® transport system to preserve DNA for standard molecular genetic analyses.
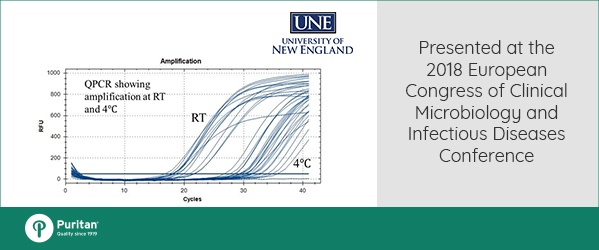
Real-time quantitative PCR assays measured concentrations of three common enteric pathogens: Escherichia coli, Shigella sonnei and Salmonella enterica, for up to 30 days at both room temperature and 4⁰C.
Conclusions were significant for all three pathogens. The yields of high quality DNA isolated from a small subsample of the medium was more than sufficient for multiple QPCR assays. This allowed the identification of many possible pathogens from a single patient sample.
Neither storage temperature or storage time had a significant effect of DNA quality. Therefore, no change in handling procedures are necessary to process patient samples for genetic analysis.
Puritan routinely works with researchers in microbiology and forensics such as the UNE team by supporting their work with product information and specimen collection devices needed for projects.
Read the entire study here.
Conclusion
For complete information on Puritan's Fecal Opti-Swab, or to request a sample, visit the Puritan Medical Products website. You'll find the Fecal Opti-Swab listed as product CB-206.