In response to an influx of inquiries we’ve received from customers, we’re responding with important CDC information about the 2019 Novel Coronavirus or 2019-nCoV.
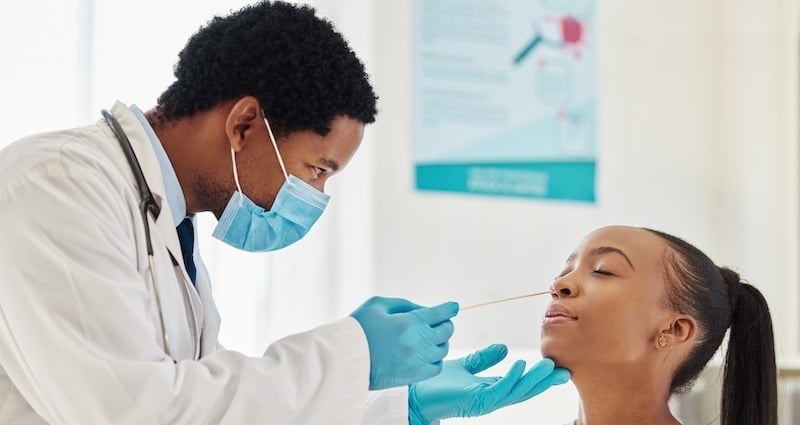
Which Swabs to Use for Coronavirus TestingSince respiratory symptoms can be a sign of a cold, the flu or, under certain conditions, the 2019-nCoV infection, laboratory tests are required for accurate diagnosis. Currently, all diagnostic testing is being conducted at the CDC.
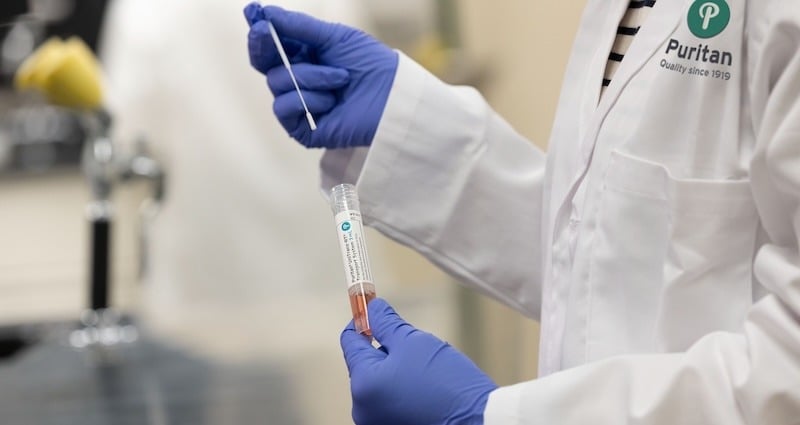
Based on CDC guidelines for clinical specimens, swabs with synthetic fibers and plastic shafts, like our flock swabs, should be used for nasopharyngeal collections when 2019-nCoV is suspected. It’s important not to use calcium alginate swabs or those with wooden shafts as they may contain materials that interfere with test results.
The tubes used should be sterile and contain 2 - 3 ml of viral transport media.
These Puritan products meet the CDC criteria:
- UT-306 – UniTranz-RT 3mL Filled Vial with Sterile Elongated Flock Swab
- UT-367 – UniTranz-RT 3mL Filled Vial with Sterile Elongated and Ultrafine Flock Swabs
- UT-302 – UniTranz-RT 3mL Filled Vial with Sterile Mini-Tip and Standard Polyester Swabs
- UT-361 – UniTranz-RT 3mL Filled Vial with Sterile Standard Polyester Swab
Please reach out to your Puritan distributor to place your order. Or feel free to contact us directly.
NOTE: Please refer to our COVID-19 portal along with the CDC and FDA websites for updated interim guidelines for collecting, handling, and testing COVID-19 specimens.