Not all swabs are created equal. Take, for instance, Puritan’s HydraFlock® and PurFlock Ultra® nasopharyngeal swabs, which are both included in the FDA's preferred swab-based types for viral specimen collection, including COVID-19 testing.
Puritan is the only U.S. manufacturer of this swab type. Unlike an ordinary cotton swab, it’s actually a highly sophisticated diagnostic tool. In fact, it’s regulated, patented, and specialized. This also means these swabs aren’t easy to manufacture on the fly.
In this article, we explore some of the characteristics that make these two styles of flock nasopharyngeal swabs ideal for viral testing. In other words, they're not your typical drugstore cotton swab.
What are the benefits of nasopharyngeal flocked swabs?
1. Tip Shape
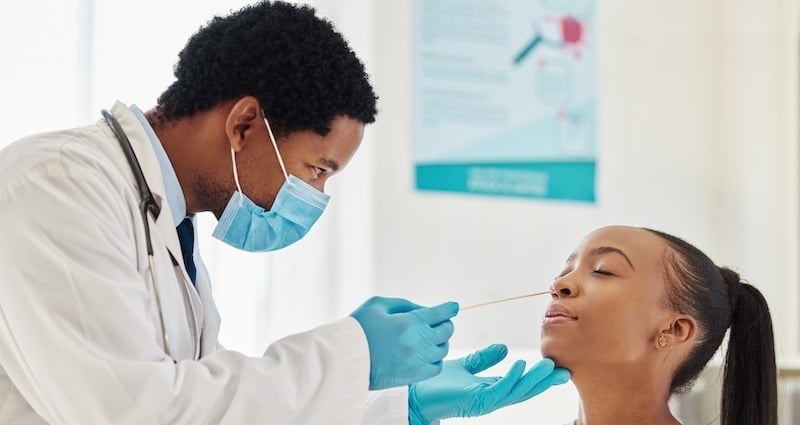
A COVID-19 specimen is typically collected from the nasopharynx, a cavity in the upper part of the throat behind the nose. A nasopharyngeal swab is specially designed to be long and thin enough to reach this area.
2. Handle Shape
The plastic handle on a nasopharyngeal swab is thin and highly flexible so it can curve easily to reach the nasopharynx and collect the specimen with minimal patient discomfort. Ideally, the handle has a breakpoint so the collection end of the swab can conveniently be broken into a vial containing universal or viral transport media.
3. Puritan's Swab Tip Material
Historically, nasopharyngeal swabs were made from nylon. Puritan’s patented swabs are made from a synthetic bi-component material that is well-suited for COVID-19 testing, because it allows for sufficient elution into the accompanying transport medium.
Not all swab materials are well suited for viral testing. Unlike drugstore swabs, nasopharyngeal swabs cannot be made with cotton or other organic materials. Why? In part because during the testing process, large amounts of virus are grown using a polymerase chain reaction (PCR). Because cotton is a plant, it has DNA that can interfere with PCR. Nasopharyngeal swabs also can’t be made with calcium alginate, commonly used on swabs for wound care, because this substance can inactivate the virus and invalidate the test.
In addition, the swab handle cannot be made of wood because this material can also interfere with test results.
4. The Exacting Requirements
As a Class I medical device, COVID-19 testing swabs must meet rigorous standards. They also need to be sterile, which adds to production time. After manufacturing, inspection and wrapping at our facility in Maine, swabs are shipped to a separate sterilization plant to complete this process. Once back at Puritan, swabs are prepared for shipment to our customers.
Looking for more? Compare Puritan’s HydraFlock® and PurFlock Ultra® nasopharyngeal swabs here.
FDA Substitutes for a Flocked Nasopharyngeal Swab
While no other swab is exactly like our HydraFlock or PurFlock Ultra nasopharyngeal swab types, the FDA and CDC have approved additional specimen collection devices with alternative tip materials to be used when flock is not available. These alternatives include some of Puritan's polyester and foam swabs.
If an approved nasopharyngeal swab is not available, alternative specimens can be collected, such as from the oropharynx or the anterior nares as well via sputum samples from patients with a productive cough.
For more information on substitute swabs and other ways to collect specimens, see current FDA guidelines for COVID-19 testing supplies under the “What If I Don’t Have” section.
Your vision, realized - Puritan Medical Products takes you from concept to commercial production seamlessly. Invest in top-quality custom swab and testing kits crafted exclusively for your needs.